Unlike ionization energy or electron affinity, the electronegativity of an atom is not a simple, fixed property that can be directly measured in a single experiment. Elements with low electronegativities tend to lose electrons in chemical reactions and are found in the lower left corner of the periodic table. Elements with high electronegativities tend to acquire electrons in chemical reactions and are found in the upper right corner of the periodic table. The most important method uses a measurement called electronegativity (represented by the Greek letter chi, χ, pronounced “ky” as in “sky”), defined as the relative ability of an atom to attract electrons to itself in a chemical compound. Conversely, the elements with the lowest ionization energies are generally those with the least negative electron affinities and are located in the lower left corner of the periodic table.īecause the tendency of an element to gain or lose electrons is so important in determining its chemistry, various methods have been developed to quantitatively describe this tendency. 
The elements with the highest ionization energies are generally those with the most negative electron affinities, which are located toward the upper right corner of the periodic table. Bonding electrons are divided equally between the bonded atoms.Nonbonding electrons are assigned to the atom on which they are located. 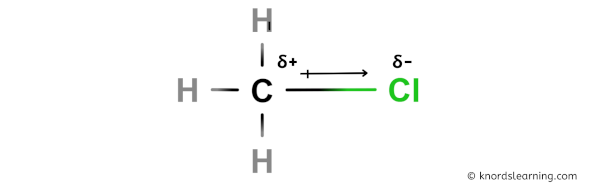
To calculate formal charges, we assign electrons in the molecule to individual atoms according to these rules: A formal charge does not represent a true charge on an atom in a covalent bond but is simply used to predict the most likely structure when a compound has more than one valid Lewis structure. The formal charge is a way of computing the charge distribution within a Lewis structure the sum of the formal charges on the atoms within a molecule or an ion must equal the overall charge on the molecule or ion. In these situations, we can choose the most stable Lewis structure by considering the formal charge on the atoms, which is the difference between the number of valence electrons in the free atom and the number assigned to it in the Lewis electron structure. It is sometimes possible to write more than one Lewis structure for a substance that does not violate the octet rule, as we saw for CH 2O, but not every Lewis structure may be equally reasonable. Note: Because of the small difference in electronegativity between carbon and hydrogen, the C-H bond is normally assumed to be nonpolar. It is often possible to rationalize chemical reactions in this manner, and you will find a knowledge of bond polarity indispensible when you start to write reaction mechanisms. This theory is borne out in practice: hydroxide ions react with chloromethane by attacking the slightly positive carbon atom in the latter. Thus, it seems reasonable that the slightly positive carbon atom in chloromethane should be susceptible to attack by a negatively charged species, such as the hydroxide ion, OH−. Now, recall that opposite charges attract. As you see, the carbon atom is shown as carrying a partial positive charge. Consider the chloromethane (CH3Cl) molecule (see page 37 of the textbook). Students often wonder why it is important to be able to tell whether or not a given bond is polar, and why they need to know which atoms carry a partial positive charge and which a partial negative charge. Van der Waals dispersion forces must be considered also.įor instance with increasing substitution $\ce$.\) Dipole moment alone can't explain the overall trends. The answer being given though is a bit of hand-waving that oversimplifies the situation. (3) Branching on carbon chain lowers the BP. (2) Increasing the number of carbon atoms (e.g. Ionic bonds > Ion-dipole interactions > H bonding > dipole-dipole interactions > Van der Waals dispersion forces. (1) Most importantly there four intermolecular forces which effect BP Yes, I think that is the right answer expected for this question. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
